Chemistry
Build an Atom
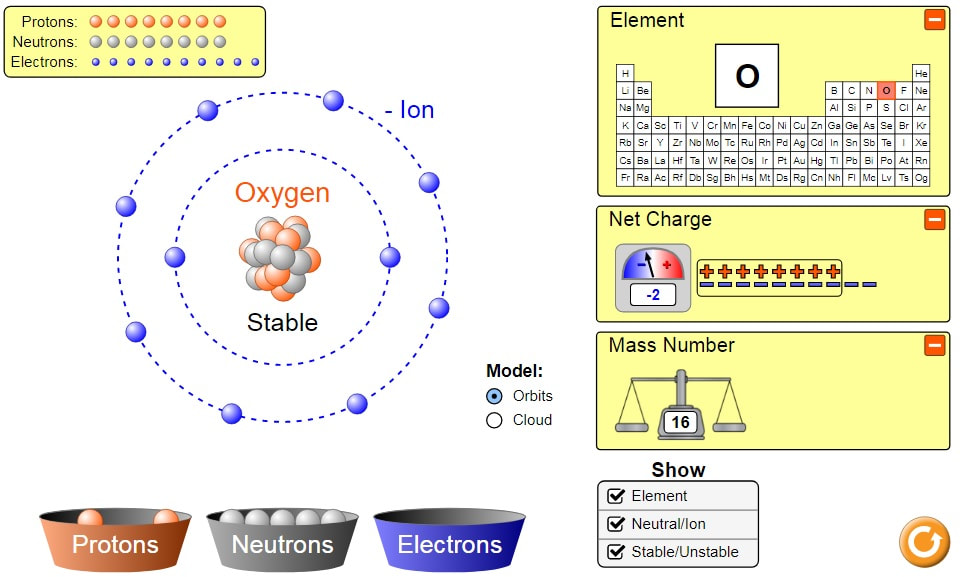
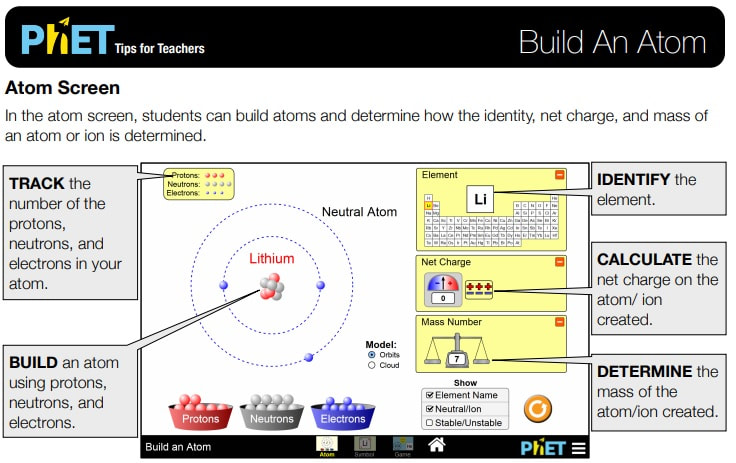
The build an atom simulation allows students to explore the structure of an atom model. By manipulating subatomic particles found in buckets below two energy shells and a nucleus, students learn how the each subatomic particle contributes to the identity and place on the periodic table of the atom, the net charge on the atom, the mass number, and the stability of the nucleus. This visual model approach allows students to see well beyond the magnification abilities of of our most powerful technology to gain an understanding of atomic structure. Students can use the simulation to design experiments, collect, analyze and interpret data, then provide evidence to support their claims.
Example prompt that an educator could use in class:
“Play until you discover what affects the mass of your atom or ion. What is a rule for determining the mass? Provide evidence to support your rule.” (Perkins, 2020, p. 46)
Once students have an understanding of atomic structure, they can challenge the game area to test and build their knowledge. Gamification has been shown to improve accessibility and engagement in the classroom (Stott & Neustaedter, 2013)
Learning goals for this simulation include:
The virtual environment is a cost effective way (it's free!), engaging, and visual way for your students to learn about subatomic particles, mass, charge, isotopes, ions, and so much more through exploration and guided inquiry. Not sure where to get started or don't have time to create an inquiry-based lab activity? There is a library of teacher-submitted editable activities at your fingertips ready to print!
Example prompt that an educator could use in class:
“Play until you discover what affects the mass of your atom or ion. What is a rule for determining the mass? Provide evidence to support your rule.” (Perkins, 2020, p. 46)
Once students have an understanding of atomic structure, they can challenge the game area to test and build their knowledge. Gamification has been shown to improve accessibility and engagement in the classroom (Stott & Neustaedter, 2013)
Learning goals for this simulation include:
- Use the number of protons, neutrons, and electrons to draw a model of the atom, identify the element, and determine the mass and charge.
- Predict how addition or subtraction of a proton, neutron, or electron will change the element, the charge, and the mass.
- Use the element name, mass, and charge to determine the number of protons, neutrons, and electrons.
- Define proton, neutron, electron, atom, and ion.
- Generate an isotopic symbol for an atom, given the number of protons, neutrons, and electrons. (PhET, 2021b)
The virtual environment is a cost effective way (it's free!), engaging, and visual way for your students to learn about subatomic particles, mass, charge, isotopes, ions, and so much more through exploration and guided inquiry. Not sure where to get started or don't have time to create an inquiry-based lab activity? There is a library of teacher-submitted editable activities at your fingertips ready to print!
Here's what the experts are saying about PhET:
- The American Chemical Society (ACS) emphasizes “One of the most important ideas about chemistry for students to learn is that what is perceived at the macroscopic level is a result of interactions at the particulate level” (American Chemical Society, 2018, p. 5). The ACS (2018) recommends that for some of these concepts and investigations, students could use computer animations and simulations such as the PhET website.
- “PhET sims are research-based tools for teaching chemistry that support the development of process skills, content learning, and affective goals in a way that is free, easily accessible, and flexible.” (Moore et al., 2014)
Sounds awesome, right? Try it out below! Or try the direct link to the simulation - Build an Atom
Need some tips to get started? YouTube has many great examples of tutorials! (Wortho's, 2020)
How to Build an Atom - Phet Simulation from Wortho's Math, Chemistry and Science Help
Professional Learning Community Extension
As mentioned above, there is a great Teacher Resource Page to learn more about the simulation or to download ready-to-print teacher-submitted activities. Note - to access the content on this page, you will need to sign up for free account.
Below are samples of the information you will find in the teacher tips PDF.
Below are samples of the information you will find in the teacher tips PDF.
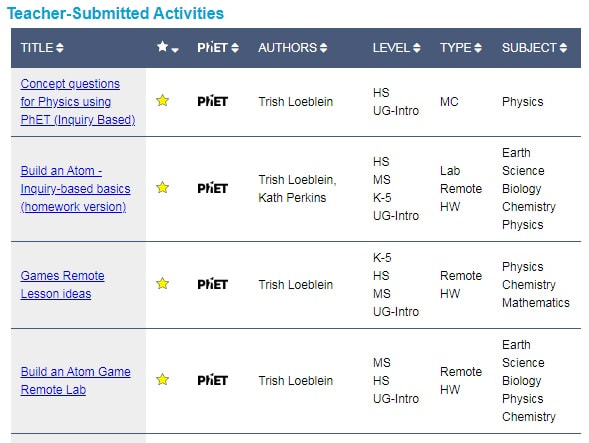
Each simulation has a collection of teacher-submitted activities. This list can be sorted by level/grade, type (demo, guided inquiry, lab, discussion, etc.), subject, creator, and also for a gold star rating which indicate high-quality, inquiry-based activities which follow the PhET design guidelines.
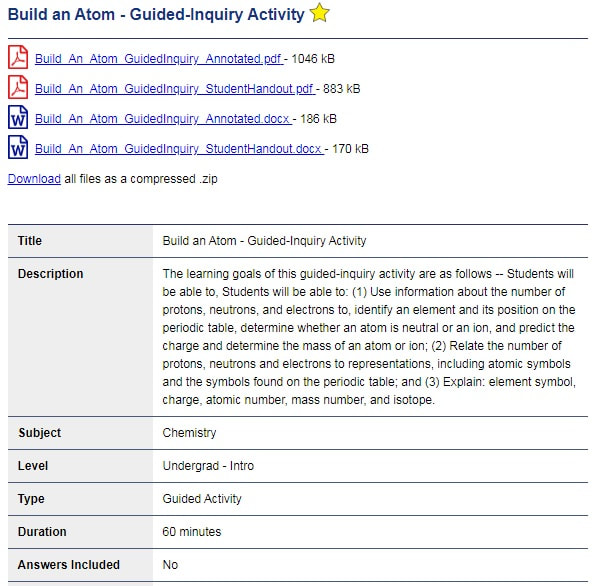
Below is an example of a teacher-submitted activity which includes student hand-outs and annotated explanations of the activity in PDF and Word formats. Other helpful information on this page to quickly determine if this activity is appropriate for you include a description, grade level, and duration.
Below is an example of a teacher-submitted activity which includes student hand-outs and annotated explanations of the activity in PDF and Word formats. Other helpful information on this page to quickly determine if this activity is appropriate for you include a description, grade level, and duration.
These activities have already been tried and tested by teachers prior to submission:
|
| ||||||||||||
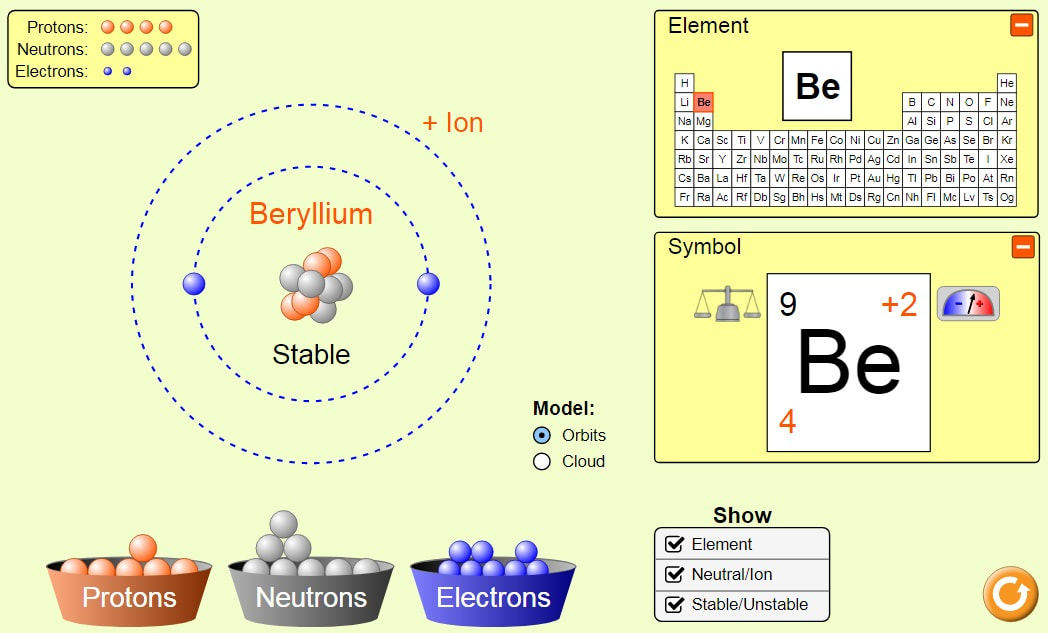
Below are sample screenshots of what students might build using the simulations:
Exploring how each subatomic particle effects the atomic number, charge, and mass number of the chemical symbol:
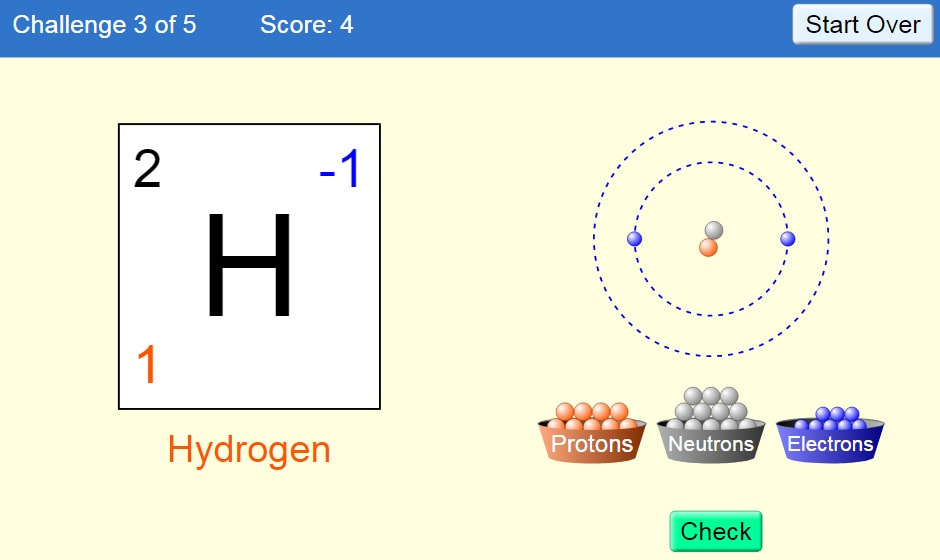
Ready to test your skills? Choose your game!
Get Started Today!
It's free! What are you waiting for? Students can access this simulation on school computers or their own devices as well! If you run into troubles or need some pre-made ideas for labs, remember to check out the Teacher Resource Page.
Go get your students started seeing the unseeable!
Go get your students started seeing the unseeable!
References:
American Chemical Society. (2018). Middle & High School Chemistry Guidelines. https://www.acs.org/content/dam/acsorg/education/policies/guidelines-teaching-mshs-chemistry/mshs-guidelines-final-2018.pdf
Moore, E. B., Chamberlain, J. M., Parson, R., & Perkins, K. K. (2014). PhET interactive simulations: Transformative tools for teaching chemistry. Journal of Chemical Education, 91(8), 1191-1197. https://doi.org/10.1021/ed4005084
Perkins, K. (2020). Transforming STEM learning at scale: PhET interactive simulations. Childhood Education, 96(4), 42–49. https://doi.org/10.1080/00094056.2020.1796451
PhET. (2021b). Build an atom. https://phet.colorado.edu/en/simulation/build-an-atom
Stott, A. & Neustaedter, C. (2013) Analysis of gamification in education. Technical Report 2013-0422-01, Connections Lab, Simon Fraser University. http://clab.iat.sfu.ca/pubs/Stott-Gamification.pdf
Wortho's Math, Chemistry and Science Help. (2020, April 3). How to Build an Atom - Phet Simulation [Video]. YouTube. https://youtu.be/wGEV7JPReC8
American Chemical Society. (2018). Middle & High School Chemistry Guidelines. https://www.acs.org/content/dam/acsorg/education/policies/guidelines-teaching-mshs-chemistry/mshs-guidelines-final-2018.pdf
Moore, E. B., Chamberlain, J. M., Parson, R., & Perkins, K. K. (2014). PhET interactive simulations: Transformative tools for teaching chemistry. Journal of Chemical Education, 91(8), 1191-1197. https://doi.org/10.1021/ed4005084
Perkins, K. (2020). Transforming STEM learning at scale: PhET interactive simulations. Childhood Education, 96(4), 42–49. https://doi.org/10.1080/00094056.2020.1796451
PhET. (2021b). Build an atom. https://phet.colorado.edu/en/simulation/build-an-atom
Stott, A. & Neustaedter, C. (2013) Analysis of gamification in education. Technical Report 2013-0422-01, Connections Lab, Simon Fraser University. http://clab.iat.sfu.ca/pubs/Stott-Gamification.pdf
Wortho's Math, Chemistry and Science Help. (2020, April 3). How to Build an Atom - Phet Simulation [Video]. YouTube. https://youtu.be/wGEV7JPReC8